
Sodium and/or volume depletion should be corrected before starting treatment with Co-Diovan. In severely sodium-depleted and/or volume-depleted patients, such as those receiving high doses of diuretics, symptomatic hypotension may occur in rare cases after initiation of therapy with Co-Diovan. Patients receiving thiazide diuretics, including hydrochlorothiazide, should be observed for clinical signs of fluid or electrolyte imbalance. This may result in hypercalcaemia.Īs for any patient receiving diuretic therapy, periodic determination of serum electrolytes should be performed at appropriate intervals. Calcium excretion is decreased by thiazide diuretics. Thiazides, including hydrochlorothiazide, increase the urinary excretion of magnesium, which may result in hypomagnesaemia. Treatment with thiazide diuretics, including hydrochlorothiazide, has been associated with hyponatraemia and hypochloraemic alkalosis. Frequent monitoring of serum potassium is recommended. Hypokalaemia has been reported under treatment with thiazide diuretics, including hydrochlorothiazide. Monitoring of potassium should be undertaken as appropriate. No dose adjustment is required in elderly patients.Ĭo-Diovan is not recommended for use in children below the age of 18 years due to a lack of data on safety and efficacy.Ĭoncomitant use with potassium supplements, potassium-sparing diuretics, salt substitutes containing potassium, or other agents that may increase potassium levels (heparin, etc.) is not recommended. Due to the valsartan component, Co-Diovan is contraindicated in patients with severe hepatic impairment or with biliary cirrhosis and cholestasis (see sections 4.3, 4.4 and 5.2). No adjustment of the hydrochlorothiazide dose is required for patients with mild to moderate hepatic impairment. In patients with mild to moderate hepatic impairment without cholestasis the dose of valsartan should not exceed 80 mg (see section 4.4). Due to the hydrochlorothiazide component, Co-Diovan is contraindicated in patients with severe renal impairment (GFR < 30 mL/min) and anuria (see sections 4.3, 4.4 and 5.2). No dose adjustment is required for patients with mild to moderate renal impairment (Glomerular Filtration Rate (GFR) ≥ 30 ml/min). This should be taken into account during dose titration.Ĭo-Diovan can be taken with or without food and should be administered with water. However, in some patients, 4-8 weeks treatment may be required. 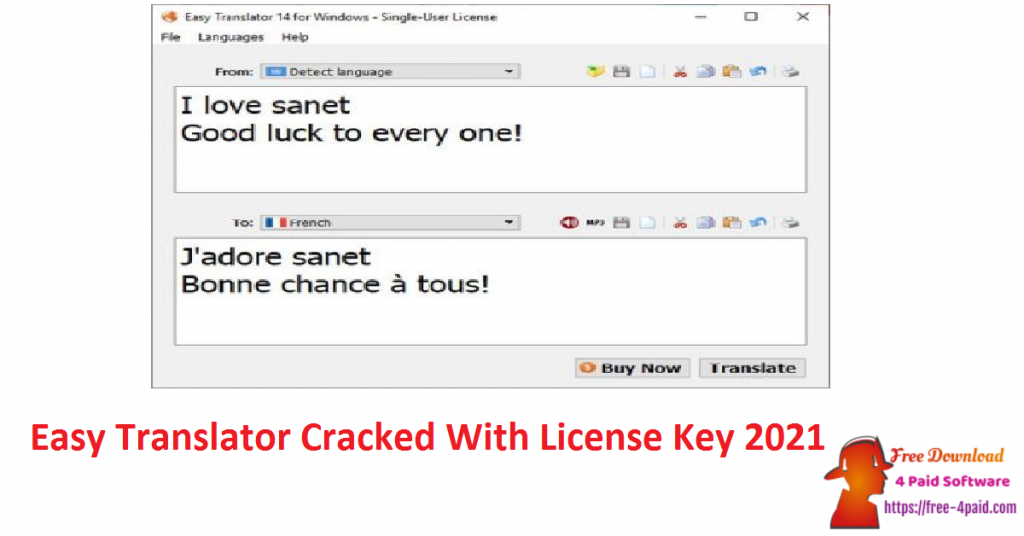
In most patients, maximal effects are observed within 4 weeks. The antihypertensive effect is substantially present within 2 weeks. The clinical response to Co-Diovan should be evaluated after initiating therapy and if blood pressure remains uncontrolled, the dose may be increased by increasing either one of the components to a maximum dose of Co-Diovan 320 mg/25 mg. When clinically appropriate direct change from monotherapy to the fixed combination may be considered in patients whose blood pressure is not adequately controlled on valsartan or hydrochlorothiazide monotherapy, provided the recommended dose titration sequence for the individual components is followed. In each case, up-titration of individual components to the next dose should be followed in order to reduce the risk of hypotension and other adverse events. Dose titration with the individual components is recommended. The recommended dose of Co-Diovan 80/12.5mg is one film-coated tablet once daily. Date of first authorisation/renewal of the authorisation 6.6 Special precautions for disposal and other handling.4.7 Effects on ability to drive and use machines.4.5 Interaction with other medicinal products and other forms of interaction.4.4 Special warnings and precautions for use.4.2 Posology and method of administration.Show table of contents Hide table of contents
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
